What two years of wearable data and a lot of scientific literature can tell us about the body’s many clocks
Estimated read time: 17 minutes
I have been tracking my heart rate variability for two years. HRV, or heart rate variability, is the slight variation in time between successive heartbeats, and it is one of the best non-invasive windows we have into the autonomic nervous system. High HRV generally signals a body in recovery mode, well-regulated and resilient. Low HRV often signals the opposite. I am perimenopausal with a Mirena IUD, which means the obvious hormonal tracking anchor, menstrual cycle phase, is largely obscured. So when I looked at a few recent months of my Fitbit data and noticed what looked like a strikingly clean correlation with lunar phases, I did what any scientist would do: I collected more data.
Two years later: the pattern did not hold.
What I found instead, and what the scientific literature illuminates beautifully, is that HRV is one of the most sensitive, most easily confounded, and most personally variable signals the body produces. It reflects hormones, stress, sleep, inflammation, aging, and yes, possibly cosmic cycles, but the story is far more complicated than any single correlation can capture. This is that story.
What HRV Actually Measures
Your heart does not beat with the mechanical regularity of a clock. The time between beats expands and contracts with every breath, every stress response, every hormonal shift. This beat-to-beat variation is heart rate variability, and it is generated by the push-pull between the two branches of the autonomic nervous system: the sympathetic (“fight or flight”) branch, which accelerates the heart, and the parasympathetic (“rest and digest”) branch, which slows it.
Higher HRV reflects a more flexible, responsive autonomic system, one where the parasympathetic brake is working well. Lower HRV can reflect chronic stress, inflammation, illness, hormonal disruption, or simply poor sleep the night before. It is not a fixed trait; it is a moment-to-moment read on physiological state. (Task Force 1996)
The most common wearable metric, RMSSD (root mean square of successive differences), captures short-term parasympathetic activity and is what Fitbit reports during sleep. Wearable HRV measurements, even high-quality ones, carry meaningful measurement uncertainty. Consumer PPG (photoplethysmography) sensors, which use light to detect blood flow through the skin, are most accurate at rest and during sleep, but can introduce systematic errors, particularly at higher HRV values. One analysis of consumer wearable data documented large errors in Fitbit HRV specifically in the higher range, suggesting possible over-correction in the device’s artifact filtering algorithm. (Altini 2024; Bent et al. 2023)
The Biology of Hormones and Autonomic Tone
Before asking whether the moon influences HRV, it is worth establishing the much better-documented driver: sex hormones. The research here is robust, mechanistically coherent, and directly relevant to anyone navigating perimenopause or using hormonal contraception.
Estrogen, Progesterone, and the Autonomic Nervous System
Estrogen, specifically estradiol, is a powerful modulator of autonomic tone. Estrogen receptors are found throughout the brain regions that regulate the autonomic nervous system, in the heart itself, and in blood vessel walls. Estrogen promotes parasympathetic (vagal) activity and suppresses sympathetic outflow. It also supports endothelial function, keeping blood vessels flexible, which indirectly supports healthy HRV by facilitating blood pressure regulation. (Zimmermann-Viehoff et al. 2022)
Progesterone works in the opposite direction. Elevated progesterone, which dominates the luteal phase of the menstrual cycle after ovulation, attenuates baroreflex sensitivity and increases sympathetic activation. The result is that HRV reliably drops in the luteal phase compared to the follicular phase in naturally cycling women. (Brar et al. 2015; Yazar and Yazici 2016)
A meta-analysis of 37 studies and over 1,000 participants confirmed this pattern: vagally-mediated HRV decreases from the follicular to the luteal phase. Two subsequent studies using multilevel models and repeated hormone measurements identified progesterone, not estradiol, as the specific driver. Within individuals, higher-than-usual progesterone predicted lower-than-usual HRV. (Ottenstein and Wiens 2020)
The pattern is not without nuance. One study found that the periovulatory phase (around ovulation) showed the most pronounced HRV dip, rather than the mid-luteal phase, suggesting that the LH surge and associated hormonal turbulence has its own autonomic signature. Other studies find inconsistency between individuals, because group-level trends do not always translate to reliable individual predictions. A person may see a clear HRV drop in the luteal phase one cycle and none in the next, driven by other stressors that overwhelm the hormonal signal. (Bouchard et al. 2023; Altini 2025)
Perimenopause: When the Signal Gets Noisier
Perimenopause, which can begin a decade before the final menstrual period, introduces erratic, unpredictable hormone fluctuations on top of the longer-term decline. This is physiologically distinct from both the regular cycling of a premenopausal woman and the stable low-estrogen state of postmenopause. (Schwarz et al. 2024)
As estrogen levels become erratic and decline, the parasympathetic protection it provided diminishes. The ANS tilts toward sympathetic dominance. The result is a body more prone to heightened stress responses, less able to recover, and with a generally lower and more variable HRV baseline. Common perimenopausal symptoms, including hot flashes, night sweats, sleep disruption, and anxiety, are themselves manifestations of autonomic dysregulation, and they create feedback loops that further suppress HRV. (Zimmermann-Viehoff et al. 2022; Schwarz et al. 2024)
One particularly notable finding: during perimenopause, women with poor sleep over extended periods sometimes showed paradoxically elevated HRV, a sign of autonomic overdrive where the nervous system is working in compensatory mode rather than genuine recovery mode. This illustrates why raw HRV numbers can be misleading without longitudinal context. (Altini 2025)
The main symptoms of perimenopause, including vasomotor instability, insomnia, blood pressure fluctuations, and cognitive changes, originate in the central nervous system’s response to hormonal disruption, not simply in the end-organs. The ANS is at the center of this transition. (Schwarz et al. 2024)
The Mirena Complication
A Mirena IUD adds another layer of hormonal complexity. It releases levonorgestrel, a synthetic progestin, directly into the uterine cavity. The systemic absorption is low, with most of the hormone remaining local, but it is not zero. Levonorgestrel is not progesterone; it is a synthetic progestin that binds to progesterone receptors but does not behave identically to the natural hormone. (Mirena prescribing information; Watson 2020)
The primary effect is local: thickened cervical mucus, thinned endometrium, and suppressed uterine activity. Ovulation is not reliably suppressed, which is why Mirena users in perimenopause continue to produce estrogen and experience hormonal fluctuations; they simply do not have bleeding as a visible marker. Mirena users may continue developing and releasing eggs and producing estrogen in the process, but without the reliable progesterone surge of a completed luteal phase. This creates a hormonal landscape that is genuinely difficult to characterize from the outside. (Watson 2020)
There is no published literature directly examining Mirena’s effects on HRV. The combination of perimenopausal hormonal variability and low-level systemic levonorgestrel creates an endocrine environment that is neither “naturally cycling” nor “post-menopausal.” It is its own moving target, and tracking HRV against a hypothesized external cycle requires first accounting for this internal hormonal noise.
The Lunar Hypothesis: What the Science Actually Says
The idea that the moon influences human physiology is ancient. The 29.5-day lunar cycle, its rough correspondence to the average human menstrual cycle (approximately 28 days), and the gravitational and electromagnetic effects of the moon have fueled speculation for millennia. In the era of wearable data and large datasets, we can examine the hypothesis more rigorously. The results are largely sobering.
What Has Been Studied
A review published in 2006 surveyed the evidence for lunar effects on human and animal physiology. It found some suggestive associations: melatonin levels appearing to correlate with lunar phase, hospital admissions for certain conditions clustering near particular moon phases, and reproductive timing in some species following lunar rhythms. The proposed mechanisms were melatonin modulation and electromagnetic radiation effects. The review was cautious about mechanistic certainty while acknowledging patterns worth investigating. (Zimecki 2006)
A study from the University of Basel gained attention for reporting shorter sleep duration, longer sleep onset, and reduced deep sleep around the full moon, measured in stringently controlled laboratory conditions. However, subsequent attempts at replication found no effect, including studies with much larger sample sizes (n = 366, n = 870). The original study’s small size (n = 33) and concerns about adequate controls have not been fully resolved. (Cajochen et al. 2013)
On the HRV question specifically: one study examining both menstrual cycle and lunar cycle effects simultaneously found that the menstrual cycle exerted strong effects on HRV, particularly around ovulation, while the lunar cycle showed only small, non-statistically-significant effects, seen at most as a marginal signal around the full moon. Even in a study designed to look for both effects in parallel, the internal hormonal signal dominated and the lunar signal was marginal at best. (Zimecki 2006; Andreatta and Tessmar-Raible 2024)
Comprehensive literature reviews and meta-analyses examining purported lunar effects on births, psychiatric admissions, emergency room visits, suicides, and behavioral outcomes have consistently found no reliable correlation between lunar phases and human biology or behavior. A review of 33 trials concluded that the overwhelming majority of evidence shows the moon has no detectable effect on human behavior, despite large proportions of healthcare professionals and the general public believing otherwise. (Roy et al. 2017)
Why the Pattern Can Look Real (And Then Disappear)
The experience of seeing a compelling lunar-HRV correlation in a few months of data, only to have it weaken with more data, is not a failure of observation. It is a textbook illustration of illusory correlation: the tendency to perceive an association that does not exist, particularly when primed to look for one. The 29.5-day lunar cycle is roughly the same duration as many biological rhythms, including HRV fluctuations driven by hormonal and seasonal cycles. When two approximately 28-to-30-day rhythms overlap in phase, they can appear correlated over short observation windows even when completely independent.
Individual HRV is highly variable, influenced by dozens of factors that themselves vary on roughly monthly timescales: stress cycles, sleep quality patterns, seasonal light exposure, exercise habits, immune activity. With enough confounders varying on similar timescales, any external approximately-29-day cycle will intermittently appear to correlate with HRV data, particularly over windows of two to four months. This is why studies finding lunar effects tend to be small, and why larger replication attempts typically fail.
This is not to say the hypothesis is absurd. Marine organisms, including bristle worms, coral, and many invertebrates, have demonstrably entrained their reproductive biology to lunar cycles, using moonlight as a reliable environmental cue. There is a credible evolutionary argument that early animals organized physiology around the predictable lunar cycle. Whether any remnant of this biological lunar clock persists in humans, detectable above the noise of modern indoor environments and behavioral variability, remains an open scientific question. As of the current evidence base, the honest answer is: we do not know, and what evidence exists is weak and inconsistent. (Andreatta and Tessmar-Raible 2024)
My Data: Two Years and a Null Result
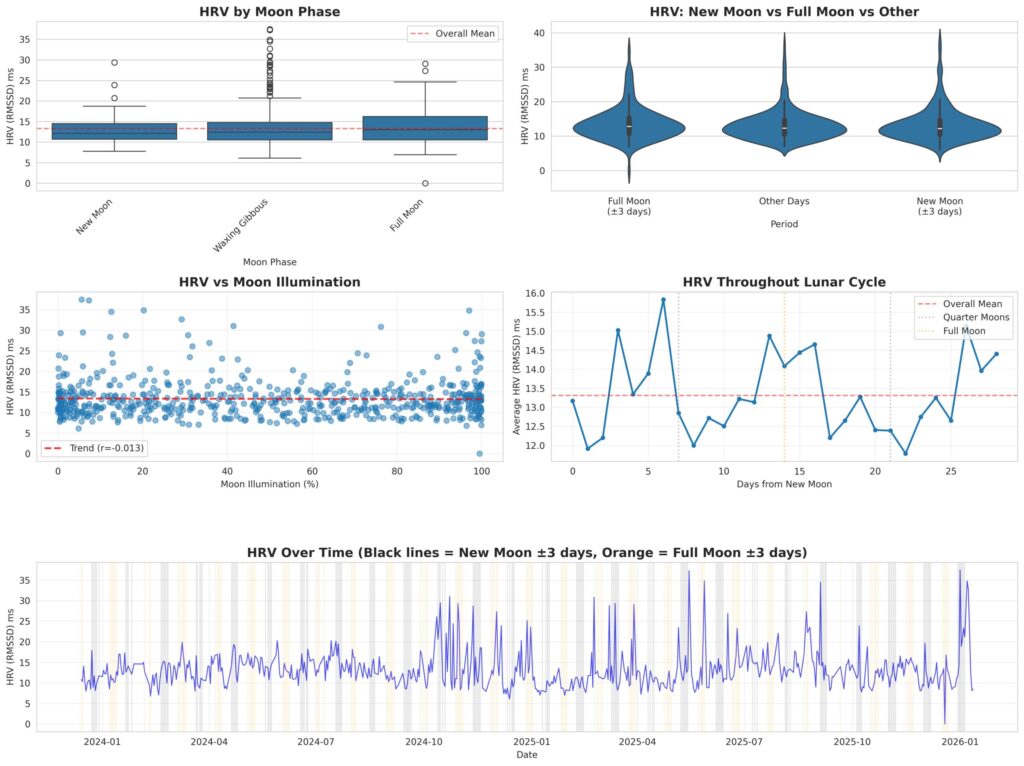
To test the lunar-HRV hypothesis, I compiled 619 days of Fitbit sleep HRV data spanning December 2023 through January 2026. Lunar phase and illumination data were calculated for each date using PyEphem version 4.2, which draws on NASA JPL ephemeris calculations, the same precision tools used in astronomy. I then ran four separate statistical tests against the HRV data.
My overall HRV across this period averaged 13.3 ms (SD 4.7 ms, range 0 to 37.5 ms).
The results across all four analyses were consistent:
Moon phase categories (new, waxing crescent, waxing gibbous, full, waning gibbous, waning crescent): One-way ANOVA, p = 0.52. No difference in mean HRV across any moon phase.
New moon vs. full moon (plus or minus 3 days each): Independent samples t-test, p = 0.63. The two groups were statistically indistinguishable.
HRV vs. moon illumination percentage (continuous correlation): Pearson r = −0.013, p = 0.75. The trend line is essentially flat, a near-zero correlation.
HRV vs. days from new moon (position in lunar cycle): Pearson r = 0.00, p = 0.99. No relationship between position in the lunar cycle and HRV.
Four tests, four null results, none approaching statistical significance. The early apparent pattern, visible in a shorter window of data, is consistent with what one would expect to find occasionally by chance in time-series data with this much natural biological variability.
What My Data Does Show
The absence of a lunar signal is not the same as an absence of all patterns. When the same dataset was analyzed for temporal structure, statistically detectable patterns emerged, just tracking different clocks than the moon.
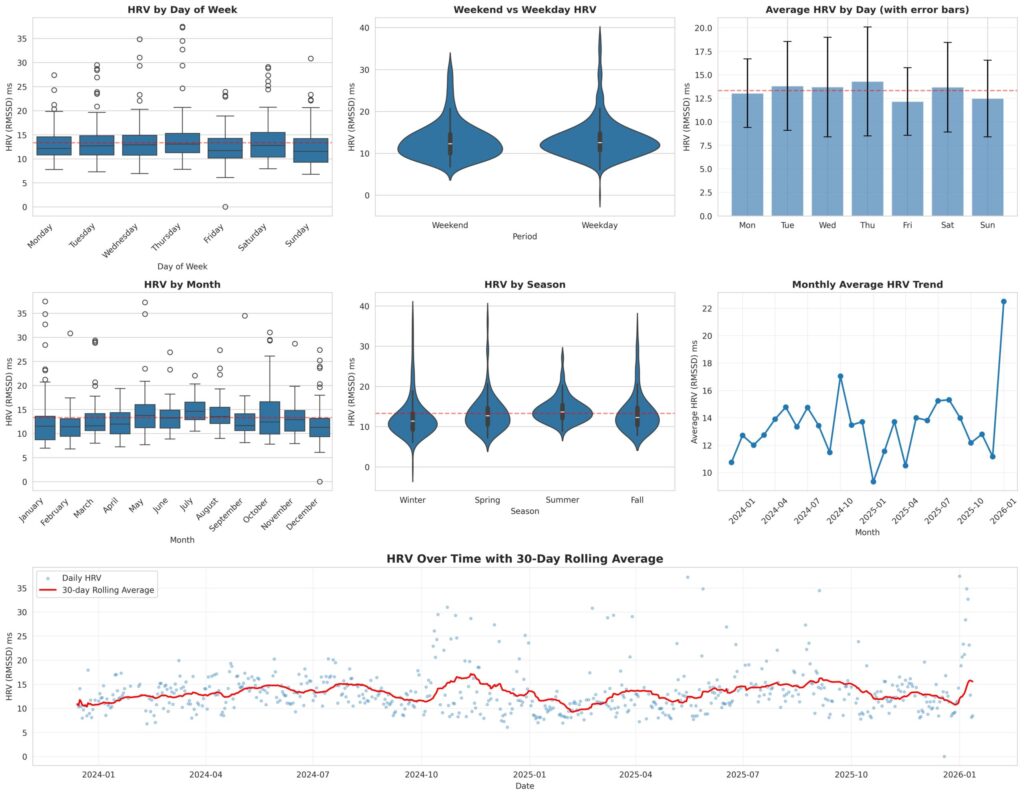
Seasonality is the most prominent pattern in my data. Summer (June through August) shows higher HRV than winter (December through February): 14.3 ms versus 12.4 ms (seasonal ANOVA p = 0.0037). The monthly breakdown shows a similar gradient, with July averaging 15.0 ms and February averaging 11.8 ms (monthly ANOVA p = 0.0023). Given the measurement noise inherent in wrist-worn PPG devices, and the absolute magnitudes involved, it would be premature to interpret these as clinically meaningful differences; they are patterns worth noting and continuing to observe rather than firm conclusions. The warm-weather elevation is consistent across May through October.
The plausible biological explanations for a seasonal pattern include increased sunlight exposure and vitamin D synthesis, warmer ambient temperatures affecting peripheral vascular tone and baroreflex function, changes in outdoor activity levels, and differences in sleep architecture across seasons. If the pattern is real, the body appears to be responding to the 365-day solar cycle, not the 29.5-day lunar one.
A day-of-week pattern also reached statistical detectability: Thursday tends to show higher HRV (14.3 ms) and Friday lower HRV (12.2 ms), with the weekly ANOVA reaching p = 0.0219. Again, the absolute difference is small and the practical meaning is uncertain given device measurement variability. The same caution applies: this is a pattern worth tracking, not a conclusion. Weekend versus weekday overall was not statistically detectable, suggesting the effect, if real, is concentrated within the workweek rather than reflecting a simple work-versus-rest divide.
These patterns are relevant to interpreting the lunar null result. My HRV data shows statistically detectable responses to seasonal and weekly rhythms but no detectable response to the lunar cycle. The nervous system does not appear to be indifferent to all cycles, only to this particular one.
What This Means for Interpreting Your Own HRV
This investigation is a useful case study in the challenges of self-quantification. Some practical takeaways:
Trends matter more than numbers. A single HRV reading is nearly meaningless in isolation. What matters is your personal baseline and directional trends over weeks. Comparing yourself to population norms is less useful than tracking your own moving average.
Context is everything. HRV without context is noise. A low reading after a difficult night, a stressful week, a viral illness, or the second half of a hormonal cycle carries meaning. A low number without context does not.
Perimenopause changes the baseline. If you are in perimenopause, your HRV baseline may be shifting on a months-long timescale that has nothing to do with any external cycle. What appears to be a response to something external may be a slow internal transition.
The absence of a visible period does not mean the absence of hormonal cycling. Mirena suppresses menstrual bleeding but does not reliably suppress ovulation or hormonal fluctuation, particularly in perimenopause. The hormonal signal is present; the visible marker is not.
Short-window correlations in personal data require skepticism. The investigation described here was worth doing, and the null result is itself informative. But a two-to-three month window of data is not sufficient to establish a real relationship in a system this noisy. More data, and ideally pre-registered hypotheses, are required before treating an apparent pattern as real.
Wearable HRV has real limitations. Consumer devices measure something real, but with meaningful error margins. Fitbit specifically has documented inaccuracies at higher HRV values. These devices are well-suited for long-term trend monitoring but should not be treated as medical-grade instruments.
What We Do Not Know
Intellectual honesty requires acknowledging what the evidence cannot yet answer.
We do not know whether there is a residual human circalunar biological clock. The evidence in marine species is clear; the evidence in humans is not. Absence of evidence in published studies is not the same as confirmed absence of effect, particularly if any effect is small and easily masked by confounders in free-living conditions.
We do not know the precise hormonal landscape of perimenopausal Mirena users. The combination has not been well characterized in autonomic research.
We do not know the detection threshold required to identify a true small lunar effect, if one exists. Most wearable studies, including two years of individual-level data analyzed here, may lack the measurement precision or sample size to detect small real effects above biological noise.
What we do know is this: the hormonal signal in HRV is large, real, and well-documented. Seasonal and weekly rhythms are detectable in personal wearable data. The lunar signal, if it exists at all in humans, is not detectable in 619 days of individual-level data using standard statistical approaches. Personal data in complex biological systems rewards careful analysis and genuine humility in equal measure.
References
Altini M. (2024). Trusting the Right Tool for the Job. Marco Altini Substack, November 22, 2024.
Altini M. (2025). Heart Rate Variability (HRV), the Menstrual Cycle, Pregnancy, and Menopause. Marco Altini Substack, March 13, 2025.
Andreatta G, Tessmar-Raible K. (2024). Time me by the moon: The evolution and function of lunar timing systems. PubMed Central PMC11316100.
Bent B, et al. (2023). Heart Rate Variability Measurement through a Smart Wearable Device. International Journal of Environmental Research and Public Health, 20(24): 7146. PMC10742885.
Bouchard P, et al. (2023). Periovulatory Subphase of the Menstrual Cycle Is Marked by a Significant Decrease in Heart Rate Variability. Biology, 12(6): 785.
Brar TK, Singh KD, Kumar A. (2015). Effect of Different Phases of Menstrual Cycle on Heart Rate Variability (HRV). Journal of Clinical and Diagnostic Research, 9(10): CC01–CC04. PMC4625231.
Cajochen C, et al. (2013). Evidence that the lunar cycle influences human sleep. Current Biology, 23(15): 1485–1488.
Danel DP, et al. (2024). Wearable-Measured Heart Rate Variability and Premenstrual Disorder Symptoms across Menstrual Cycle. medRxiv preprint. doi:10.1101/2024.10.27.24316196.
Feehan LM, et al. (2020). Accuracy of Fitbit Devices: Systematic Review and Narrative Syntheses of Quantitative Data. JMIR mHealth and uHealth, 8(8): e18694.
Ottenstein V, Wiens S. (2020). Menstrual Cycle Changes in Vagally-Mediated Heart Rate Variability Are Associated with Progesterone: Evidence from Two Within-Person Studies. Journal of Clinical Medicine, 9(3): 617. PMC7141121.
Roy A, Biswas T, Roy AK. (2017). A Structured Review of Relation between Full Moon and Different Aspects of Human Health. SM Journal of Biometrics and Biostatistics, 2(1): 1007.
Schwarz KG, et al. (2024). Autonomic nervous system dysfunction throughout menopausal transition: A potential mechanism underpinning cardiovascular and cognitive alterations during female ageing. International Menopause Society.
Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. (1996). Heart rate variability: standards of measurement, physiological interpretation and clinical use. Circulation, 93(5): 1043–1065.
Watson L. (2020). Mirena and Your Hormones. drlisawatson.com.
Yazar S, Yazici M. (2016). Impact of Menstrual Cycle on Cardiac Autonomic Function Assessed by Heart Rate Variability and Heart Rate Recovery. Medical Principles and Practice, 25(4): 374–377. PMC5588411.
Zimecki M. (2006). The lunar cycle: effects on human and animal behavior and physiology. Postepy Higieny i Medycyny Doswiadczalnej, 60: 1–7. PubMed 16407788.
Zimmermann-Viehoff F, et al. (2022). Heart rate variability as a function of menopausal status, menstrual cycle phase, and estradiol level. Physiological Reports. PMC9127980.
Author note: This article synthesizes peer-reviewed research and the author’s own two-year dataset (n = 619 days). Statistical analyses were performed using Python (scipy.stats); lunar phase calculations used PyEphem v4.2 with NASA JPL ephemeris data. All factual claims are attributed to cited sources. Where evidence is limited or conflicting, this has been noted explicitly. This article is written for informational purposes and does not constitute medical advice.

Leave a Reply